DHA’S INFLUENCE ON THE GUT
The many benefits of omega-3s have long been understood, but in many cases the mechanism by which they had these positive effects has been less clear. Recent research is beginning to show us that at least some of these effects could be due their influence on the microorganisms that live in our guts. The human gut microbiome is the collection of microorganisms (microbiota) in the human digestive tract, and is an interconnected ecosystem of bacteria, yeasts and other organisms such as protists, archaea and viruses. It is the largest reservoir of microorganisms in the human body and a contributes much of the enzymatic activity essential for human digestion and physiology.
The microbiota and its human host are an example of mutualistic symbiosis, where both sides of the partnership gain a benefit. Several factors influence the composition of the microbiota, both in the types of organisms present and their abundance, and these make our total gut microbiota profiles very host-specific. Diet is one of the most influential factors impacting the composition and stability of the gut microbiota, but other factors such as age, gender, body mass, lifestyle choices, and immune function can have a large impact alongside environmental factors linked to, for example geographical location. The use of antibiotics, drugs, alcohol, and probiotics can also have a significant impact on the microbiota.
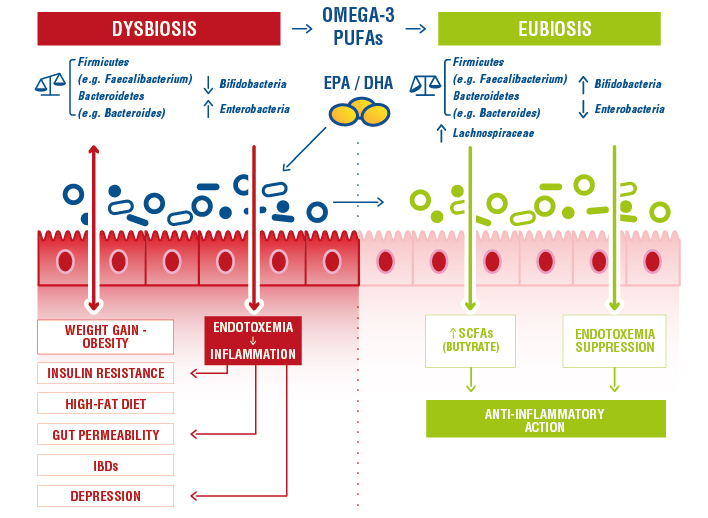
In conditions of dysbiosis, there is a change in the composition or stability of the bacterial populations of the intestine such that the normal healthy symbiotic relationship is impaired. This can be caused by many factors such as antibiotic use, illness or poor diet, but can lead to longer-term health consequences. Dysbiotic microbiota can be associated with metabolic disorders such as type 2 diabetes, obesity or cardiovascular diseases, and certain components of an unbalanced microbiota have been associated with chronic inflammatory bowel diseases such as Crohn’s disease or ulcerative colitis, and also with the development of allergies and colorectal cancer.
Restoration of a normal microbiota and reduction of gut inflammtion thanks to algal oil
Some metabolic disorders of the host are thought to be associated with a composition of the gut microbiota with an abnormal influence on inflammation. Different bacteria in the gut can modulate immune functionality and can play pro- and anti-inflammatory roles. Thus, the composition of the microbiota contributes to the host’s level of resistance to infection and susceptibility to inflammatory diseases. Omega-3 PUFAs exert significant effects on the gut environment by modulating the composition of the gut microbiota, promoting an increase in those groups which produce anti-inflammatory compounds, like short-chain fatty acids (1,2).
Antibiotics can be particularly disruptive to the microbiota, especially in early life, and omega-3s can alleviate the problems that these may cause, and reduce the risks of problems later in life caused by the imbalance such as obesity (2) .
In a recent study, Yang et al. studied the effect of DHA-rich algal oils on dysbiosis caused by antibiotic treatment. Algae oil treatment (500 mg kg-1 day-1) restored the balance of the gut microbiota by increasing the number of short-chain fatty acid-producing bacteria, such as Ruminococcus and Blautia and significantly reduced levels of pro-inflammatory cytokines, including interleukin 6, interleukin 1β and tumour necrosis factor α, in the colon. They conclude that supplementation with algal oil could alleviate inflammation and promote mucosal healing, which suggests that DHA-rich algal oil could be used to protect against antibiotic-induced alteration of the gut microbiota and metabolic dysbiosis (3) .
Figure 1 : changes in the gut microbiota following intake of omega-3 oils learning to reduction in dysbiosis (3).
DHA influence the gut-brain axis
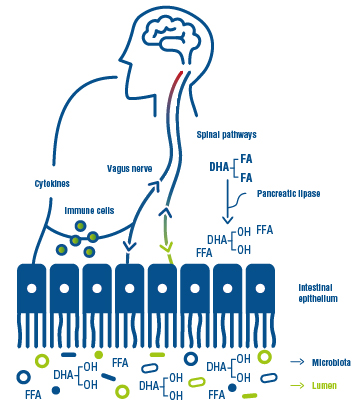
DHA’s important structural and functional role in maintaining proper brain and nervous functions is well known, but the gut microbiota can also, surprisingly, have an effect on the function of the brain via the gut-brain axis. There has been recent interest in the possible interaction between several brain problems and intestinal microflora; emotional disorders such as depression, anxiety and stress have been demonstrated to show a correlation with gut microbiota alterations (4). Bacterial colonization of different species can potentially alter brain functions, and in turn, the central nervous system is speculated to indirectly influence the gut microbial composition (5).
Figure 2 : interactions between the gut and brain, with DHA playing a role un both microbiota and nerve composition.
Dietary interventions with DHA may have beneficial effects on behavioural and neurophysiological disorders not only via direct action on the brain but also due to the alteration of the microbial composition of the gut and an indirect action via the blood-gut axis. Higher circulating levels of DHA has certainly been show to correlate with better microbiota diversity (6).
Interestingly the differences in prevalence of some mental disorders between the sexes may also be linked to differences in the microbiota. Studies in mice, for example show that DHA can lead to reductions in symptoms of anxiety and depression in socially isolated males, but not in females, and this is linked to the microbiome (7).
Chronis inflammatory bowel disease and Colorectal Cancer
Chronic inflammatory diseases of the gut including ulcerative-colitis and Crohn’s disease are associated with gut microbiome dysbiosis and there is now some evidence to suggest that may also be linked to imbalances in omega-3 and omega-6 metabolism, especially their transformation into either anti-inflammatory or inflammatory cytokines (8) . While some patients report improved symptoms from taking omega-3s, the evidence for their efficacy in management of these diseases is still unclear (9).
Nonetheless, it is clear that inflammation observed in ulcerative-colitis and Crohn’s disease increases the risk of colorectal cancer (CRC) above that of the general population. The pro-inflammatory context of these diseases promotes virtually all steps of colon carcinogenesis and patients with CRC show an increased level of circulating proinflammatory mediators (IL-6, IL-1β, IL-8 and TNFα).
Epidemiological and clinical studies suggest beneficial effects of anti-inflammatory omega-3 polyunsaturated fatty acid intake on the risk of CRC, and it is also being considered as having a role in treatment (10). DHA’s role might be a consequence of its ability to regulate the production of pro-inflammatory mediators in cancer cells and/or host cells changing the systemic inflammatory status or that of the tumour microenvironment.
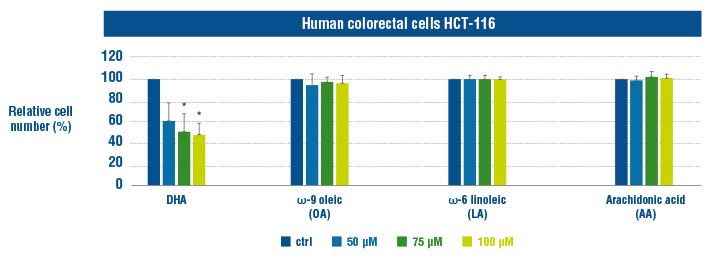
Fluckiger et al. (12) have demonstrated that DHA treatment induced inhibition of tumour growth and activation of apoptosis in colon cancer cells through a TNFα pathway. The effect of DHA was evaluated on growth of human colon cancer cells (HCT-116 and HCT-8 cells) using Cyquant assay and compared with ω-9 oleic (OA), ω-6 linoleic (LA) and arachidonic (AA) acids. DHA treatment (50, 75 and 100 μM) for 48 h reduced cancer cell number in a dose-dependent manner while OA, AA acid and LA treatment did not in comparison with the control (CTRL) treatment (11).
The microbial population of our guts has an important effect on many aspects of our health and well-being. Antibiotics, poor diet and other elements of our lifestyles such as smoking and alcohol consumption can disturb the delicate balance of species that inhabit our intestines.
Omega 3s have a proven role in restoring and maintaining a healthy intestinal balance, and reducing intestinal inflammation. DHA promotes the development of short-chain fatty acid-producing bacteria while reducing pro-inflammatory molecules. These microbial metabolites act as a messenger in the brain-gut axis and are correlated with a reduction in anxiety and depression.
In addition to acting on the microbiota, DHA has a potentially curative role in cases of colon cancer, the risk of which is increased by chronic inflammation, by significantly reducing the number of cancerous cells.
Bibliography :
- Costantini, L., Molinari, R., Farinon, B. & Merendino, N. Impact of Omega-3 Fatty Acids on the Gut Microbiota. Int. J. Mol. Sci. 18, E2645 (2017).
- Yawei Fu , Yadong Wang , Hu Gao, DongHua Li, RuiRui Jiang, Lingrui Ge, Chao Tong, and Kang Xu. Associations among Dietary Omega-3 Polyunsaturated Fatty Acids, the Gut Microbiota, and Intestinal Immunity. (2021)
- Kaliannan, K., Wang, B., Li, X.-Y., Bhan, A. K. & Kang, J. X. Omega-3 fatty acids prevent early-life antibiotic exposure-induced gut microbiota dysbiosis and later-life obesity. Int. J. Obes. 2005 40, 1039–1042 (2016).
- Yang, C. et al. Algal Oil Rich in Docosahexaenoic Acid Alleviates Intestinal Inflammation Induced by Antibiotics Associated with the Modulation of the Gut Microbiome and Metabolome. J. Agric. Food Chem. (2021) doi:10.1021/acs.jafc.0c07323.
- Taylor, A. M. & Holscher, H. D. A review of dietary and microbial connections to depression, anxiety, and stress. Nutr. Neurosci. 23, 237–250 (2020).
- Jin, J., Jin, Q., Wang, X. & Akoh, C. C. High Sn-2 Docosahexaenoic Acid Lipids for Brain Benefits, and Their Enzymatic Syntheses: A Review. Engineering 6, 424–431 (2020).
- Menni, C. et al. Omega-3 fatty acids correlate with gut microbiome diversity and production of N-carbamylglutamate in middle aged and elderly women. Sci. Rep. 7, 11079 (2017).
- Davis, D. J. et al. Sex-specific effects of docosahexaenoic acid (DHA) on the microbiome and behavior of socially-isolated mice. Brain. Behav. Immun. 59, 38–48 (2017).
- Scoville, E. A. et al. Serum Polyunsaturated Fatty Acids Correlate with Serum Cytokines and Clinical Disease Activity in Crohn’s Disease. Sci. Rep. 9, 2882 (2019).
- Turner, D., Shah, P. S., Steinhart, A. H., Zlotkin, S. & Griffiths, A. M. Maintenance of remission in inflammatory bowel disease using omega-3 fatty acids (fish oil): a systematic review and meta-analyses. Inflamm. Bowel Dis. 17, 336–345 (2011).
- Volpato, M. & Hull, M. A. Omega-3 polyunsaturated fatty acids as adjuvant therapy of colorectal cancer. Cancer Metastasis Rev. 37, 545–555 (2018).
- Fluckiger, A. et al. Inhibition of colon cancer growth by docosahexaenoic acid involves autocrine production of TNFα. Oncogene 35, 4611–4622 (2016).